|
Ø
��Ʒ����
������ �绰��010-84369508 |
Ø
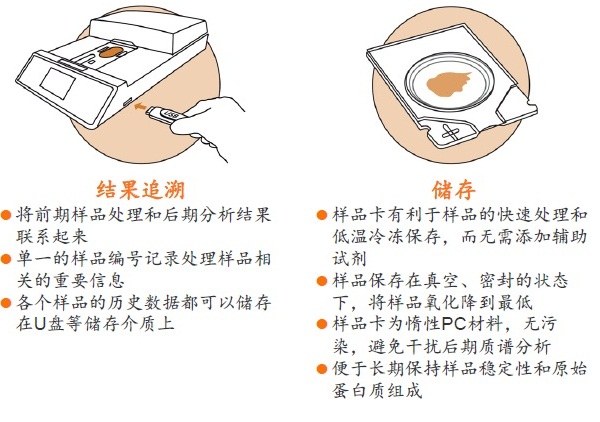
Stabilizor T1 ������Ʒ�ȶ���
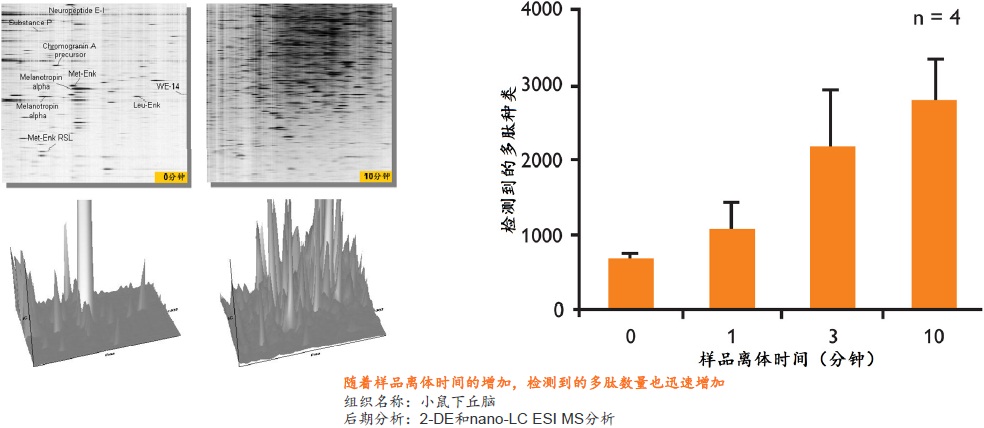
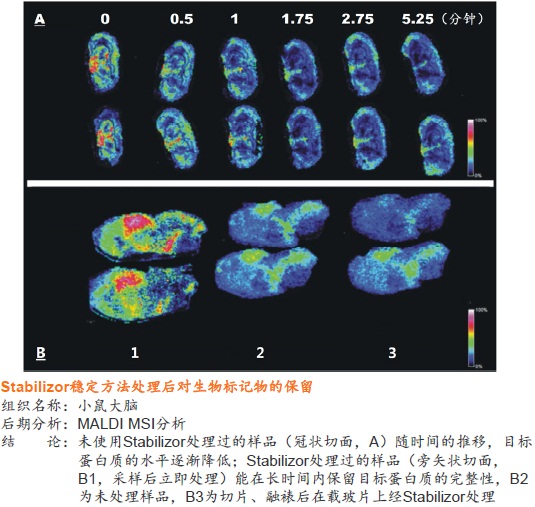
�ڵ��������о��У�Ҫ�õ�ȷ�ɿ��Ľ�����ܴ�̶���ȡ������Ʒ��ǰ����������������Ʒ���ȶ��ԣ�����Ʒ�Ƿ�ӳĿ�군�����ڻ����ڵ�ԭʼ״̬����Ϊ��Ҫ��һ����Ʒ�ӻ�����룬����ƽ�ⱻ���ƣ�����ijЩ�����ʻ���ģ����彵����һ���ܿ��ٵĹ��̡�����µ����ʵ����ࡢ���������ο��ٱ仯��ʹ�ü��IJ��ֵ����ʻ��Ķ�ʵ�����Ǹ߷�ȵ����ʵĽ��������Ƭ�Σ���������Ʒ��ԭʼ�����ʣ�����ͼ����
��ϸ������֯����ĵ��������У�������Ҫ�������ܡ�����о�����ͼ�ֵ�ĵ�����ͨ����һЩ������С���ȶ��Բ�ĵͷ�ȵ����ʻ���ġ����ŵ����ʼ�⼼���ķ�չ����ⷽ����������Խ��Խ�ߣ�Ҳ����Ʒǰ������������˸��ߵ�Ҫ������ڴ�����Ʒ�����б�����Щ�����ʻ���ľͱ��Խ��Խ��Ҫ��
��˲���һ�ֿ��١���Ч����Ʒ�ȶ�����������������Ʒ�ɼ����ȶ�֮��ļ��ʱ�䲢����Ʒǰ�������̱��������ڵ������о�������Ҫ���塣����������Ʒǰ���������ڻ�ø����������ݣ�������ǿ��������Ŀɿ��ԺͿɱ��ԡ�
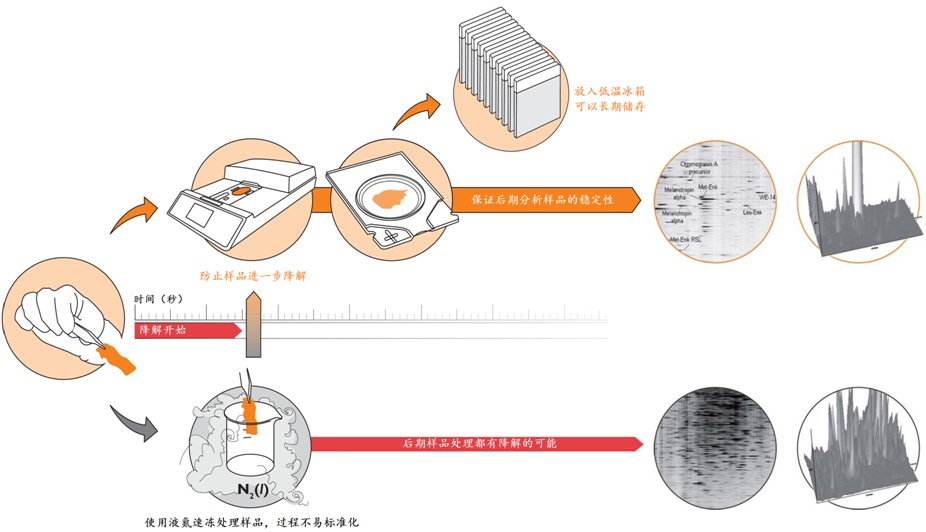
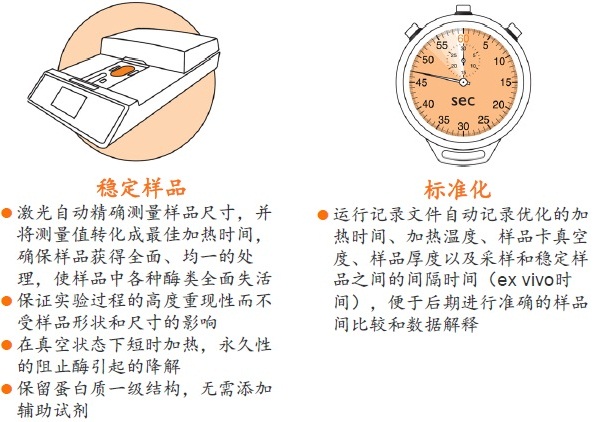
Stabilizorϵͳ�����Denator��˾��ר��������Ʒ�ȶ���������ϵͳ����Stabilizor T1������Maintainor��Ʒ�������ڽ�����������彵������⡣Stabilizorϵͳ�ܿ��١����ȡ�ȷ�Ľ��������ݸ���Ʒ���ƻ��������е�������������������Ĺ����ļ������ƻ������ʵĶ��������Ͻṹ������һ���ṹ�������ԡ����ֲ������ʧ��ЧӦ����Ʒ�еĸ���ø��ʧ��Ӷ������������彵��ij̶Ƚ�����͡������������������Ӹ��������Լ���Ҳ�����ڷ����ĸ������ؽ�������͡�
ʹ��Stabilizorϵͳ�������Ӹ��������Լ����������Ե�ȥ������ø������ø�Ļ��ԣ���ֹ�����ʽ��ⷢ�������͵�������ĸ����ԣ����ٸ߷�ȵ����ʽ���Ƭ�εĸ��ţ����ӷ����ͷ��־�����Ҫ��ֵ�ĵ�����/������������Ŀ����ԡ�Stabilizor������Ʒ���е��ȶ��ԡ������Ϳ����ԣ����õ��������ķ������ݡ�
Һ���ٶ����ø���Ƽ���Ŀǰ�㷺ʹ�õ���ֹ��Ʒ����ı������������ַ������ƻ�ϸ��Ĥ��ϸ���ṹ��ʹ����Ʒ�ڻ���������ε����ʵ�ø�ֲ��ø��㷺�����¸��൰���ʻ���������ʽ�����ı䡣�������ַ������п����Ժ�ѡ���ԣ���ȫ�����ֹ��Ʒ��ø��Ļ��ԣ����ӵ��Լ�������ź�����Ʒ�����������Լ�����һ�����ԡ�
ʹ��Stabilizorϵͳ�����ƣ�
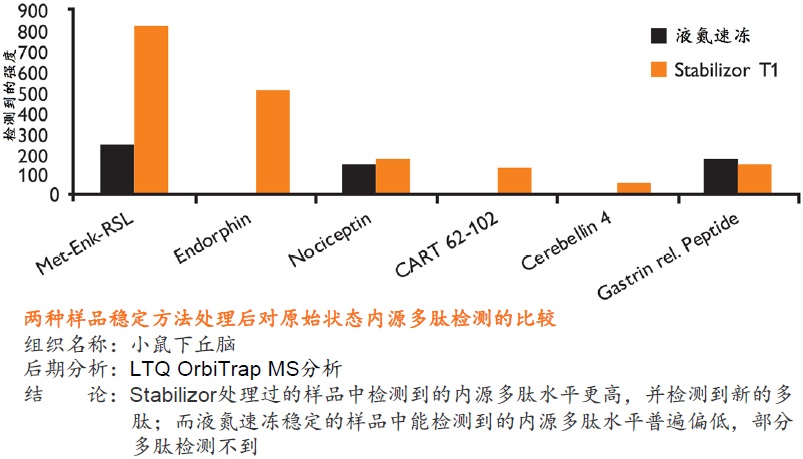
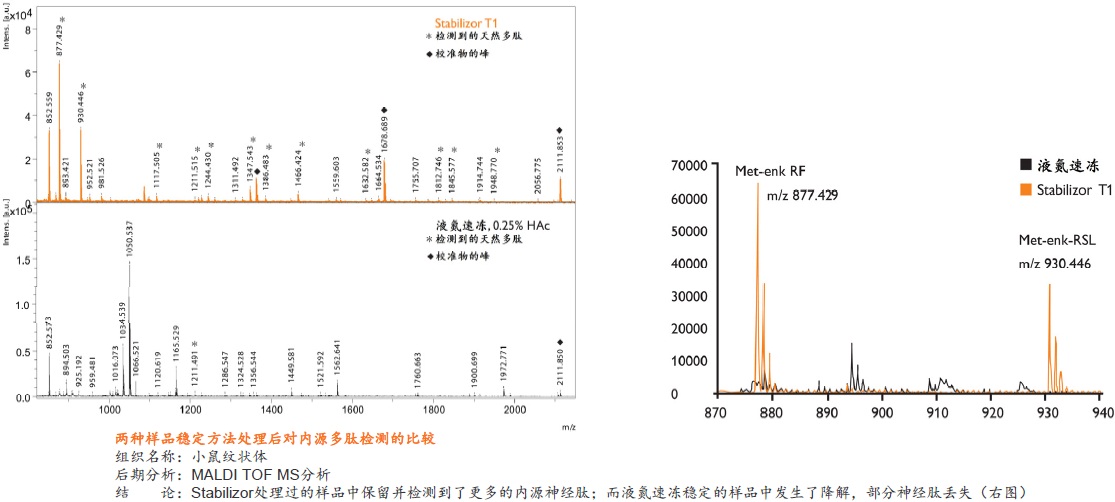
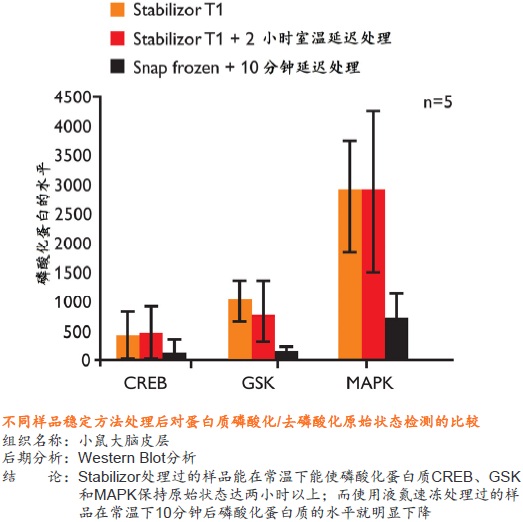
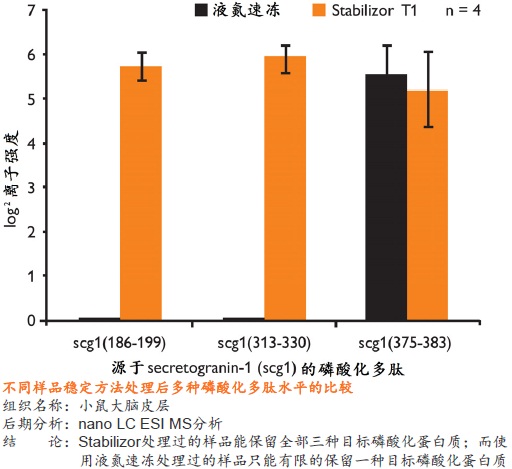
�� ��һ�ִ���������Ʒ�ȶ�����������ʹ�ø��ź��ڷ�����ø���Ƽ� �� ����ʱ��̣�һ����һ�������ڼ��ɽ���Ʒ�еĸ���ø��ʧ�� �� �����ڱ�����Ʒ�ڻ����ڵ����ʵ����ữ��������������ε���ʵ״̬���������ȶ�������ĵͷ�ȵ����� �� ��������Ʒ�еͷ�ȵ����ʻ�����ܸ߷�ȵ����ʽ���Ƭ�εĸ��ţ������ڷ����µĵ����ʡ������Լ����ǵ�������ʽ
�� �����Ե�ȥ������ø������ø�Ļ��ԣ���Ʒ���ȶ������ڳ����½����Ʊ����������赣��ø�Ե����ʡ����ĵĽ����ı����ǵ�������ʽ �� ���͵�������ĸ��ӳ̶ȣ���С��̬��Χ�����ӷ����ͷ��־�����Ҫ��ֵ�ĵ����ʻ������������Ŀ�����
�� ʹ�÷��㣬�ڲ�����ͬʱ�����ȶ���Ʒ��������Ʒ���彵���ʱ��
�� ��Ʒ�ɼ��ʹ���Ĺ��̸߶ȱ�����ʹ����������ӿɿ������ݸ��߿ɱ��ԣ����ڽ��бȽϵ��������о�
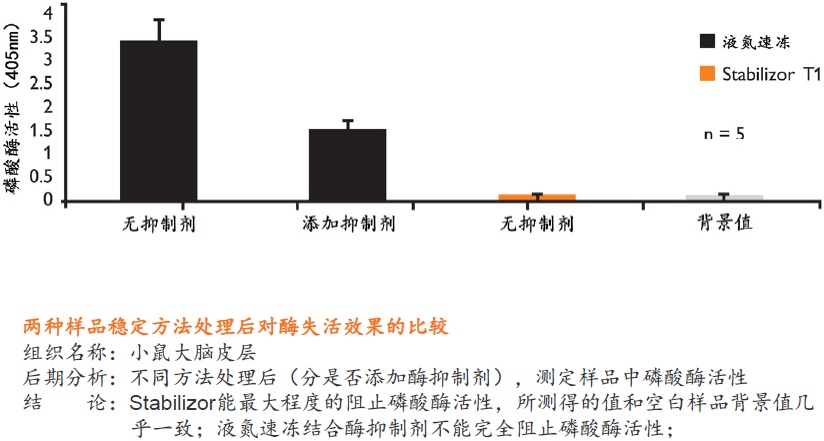
�� ʹ��MALDI-MSI���Ŀ�군���ʻ���ģ�Stabilizor��Ŀǰ��Ϊ��Ч����Ʒ�ȶ�����
����������
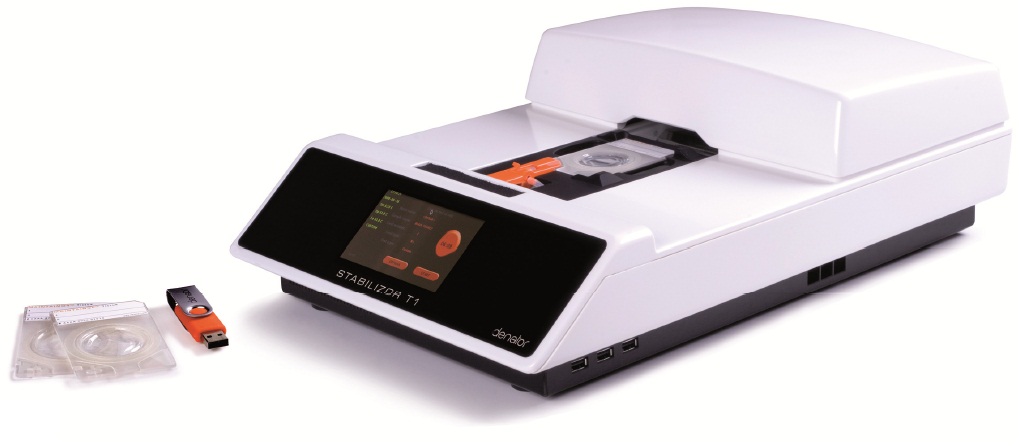
Stabilizor T1����
�� 4.3Ӣ���ɫ������ʾ�� �� ����Ʒ��ʩ�����ѹ����Χ��10��100mbar �� ����������45�ֱ� �� ��Ʒ�ߴ��������ּ���CLASS 1��ɫ���� �� ���ô�������1 GHz��86���ڴ�Ϊ1GB �� ����ϵͳ��Linux��Դ���� �� ����USB�ӿ� �� �ⲿ�ߴ磨L��W��H����465��306��143mm �� ���أ� 6.7kg �� ���ʣ����Ϊ350�ߣ�����ʹ��Ϊ70�ߣ�����Ϊ20�� �� ��Դ��100-240V/50-60Hz �� ʹ�û����¶ȣ�10��40�� �� ʹ�û���������ʪ�ȣ�80%�������ᣩ Maintainor��Ʒ��
�� �ⲿ�ߴ磨L��W��H����85��54��2mm �� ��Ʒǻ�ߴ磺��7mm��ֱ��33mm �� ʹ�û���������ʪ�ȣ�85%�������ᣩ �� ���ʣ�PC�����ط���Ϳ�� �� �����¶ȣ�-80��100 ��
Stabilizorϵͳ�����ڽ�ʾ�����ĵ������顢����������ữͼ��
������Ϣ��
ʹ��Stabilizor������Ʒ�������IJ�������Loss of correlations among proteins in brains of the Ts65Dn mouse model of Down syndrome. Ahmed et al., J. of Proteome research. 2012. Just accepted Read more Sample Preparation Techniques for the Untargeted LC-MS-Based Discovery of Peptides in Complex Biological Matrices. Finoulst et al., J. Biomedicine and Biotechnology. 2011. Read more Extensive Characterization of Tupaia belangeri Neuropeptidome Using an Integrated Mass Spectrometric Approach. Petruzziello et al., J. Proteome Res. 2011, ahead of print. Read more Effect of Thaw Temperatures in Reducing Enzyme Activity in Human Thyroid Tissues. Kokkat et al., Biopreservation and Biobanking. 2011, ahead of print. Read more Thermal stabilization of tissues and the preservation of protein phosphorylation states for two-dimensional gel electrophoresis. Smejkal et al., Electrophoresis 2011, Volume 32, pages 2206�C2215. Read more Biomarkers of disease and post-mortem changes-heat stabilization, a necessary tool for measurement of protein regulation. Kultima et al., Journal of Proteomics, 2011 In press. Read more Benefits of heat-treatment to the protease packed neutrophil for proteome analysis: Halting protein degradation. Kennedy et al., Proteomics 2011, 11, pages 2560�C2564 Read more Neuropeptide profiling of the bovine hypothalamus: Thermal stabilization is an effective tool in inhibiting post-mortem degradation. Colgrave et al., Proteomics 2011, 11, 1264-1276 Read more Methodology and technology for stabilization of specific states of signal transduction proteins. Bor��n M., Methods Mol Biol. 2011;717:91-100 Read more Preserving protein profiles in tissue samples: Differing outcomes with and without heat stabilization, M Ahmed et al, Journal of Neuroscience Methods. 2011 Mar 15;196(1):99-106 Read more Clinical application for the preservation of phospho-proteins through in-situ tissue stabilization, CB Rountree et al, Proteome Science, 2010; 8: 61 Read more Stopping the clock on proteomic degradation by heat-treatment at the point of tissue excision. Richard J. A. Goodwin et al. Proteomics, Volume 10, Issue 9, pages 1751-1761, No. 9 May 2010 Read more Mass Spectrometric Imaging for Biomedical Tissue, Chughtai, K. and. Heeren, R.M.A, Analysis, Chem. Rev., 2010, 110 (5), pp 3237-3277. Read more Neuropeptidomic analysis of the embryonic Japanese quail diencephalon, Scholz, B., H. Alm, et al., BMC Dev Biol 10: 30. Read more The use of neuroproteomics in drug abuse research, M Lull et al, Drug and Alcohol Dependence, Volume 107, Issue 1, 1 February 2010, Pages 11-22 Read more Impact of temperature dependent sampling procedures in proteomics and peptidomics - A characterization of the liver and pancreas post mortem degradome, B Scholz et al. Molecular and Cellular Proteomics, M900229-MCP200, January 28, 2010 Read more Banking of clinical samples for proteomic biomarker studies: A consideration of logistical issues with a focus on pre-analytical variation, Jackson, D.H., and Banks, R.E., Proteomics - Clinical Applications 2010 4:3, 250-270 Read more A report on the ESF workshop on quality control in proteomics, L Martens. Mol. BioSyst., 2010, 6, 935-938 Read more Assessing the use of thermal treatment to preserve the intact proteomes of post-mortem heart and brain tissue, AA. Robinson et al. Proteomics., Article 2009 Oct;9(19):4433-44 Read more Preserving the yeast proteome from sample degradation, J. Grassl et al. Proteomics., Proteomics. 2009 Oct;9(20):4616-26. Read more Development and evaluation of normalization methods for label-free relative quantification of endogenous peptides. K. Kultima, Molecular and Cellular Proteomics, October 1, 2009, 8, 2285-2295. Read more A Quantitative Peptidomic Analysis of Peptides Related to the Endogenous Opioid and Tachykinin Systems in Nucleus Accumbens of Rats Following Naloxone-Precipitated Morphine Withdrawal U. Rossbach et al. J. Proteome Res., Proteomics. 2009 Oct;9(20):4616-26 Read more Heat Stabilization of the Tissue Proteome: A New Technology for Improved Proteomics, M. Svensson et al. J. Proteome Res., 2009, 8 (2), pp 974-981 Read more Sample preparation issues for tissue imaging by imaging MS, Kaletas, B. K., I. M. van der Wiel, et al. Proteomics 9(10): 2622-33 Read more Protein and peptides in pictures: imaging with MALDI mass spectrometry, RJ. Goodwin et al. Proteomics., 2008 Sep;8(18):3785-800 Read more The significance of biochemical and molecular sample integrity in brain proteomics and peptidomics: stathmin 2-20 and peptides as sample quality indicators. K. Sköld et al. Proteomics. 2007 Dec;7(24):4445-56 Read more Neuropeptidomics: MS Applied to the Discovery of Novel Peptides from the Brain M. Svensson et al. Anal. Chem., 2007, 79 (1), pp 14-21 Read more Neuropeptidomics: expanding proteomics downwards. M. Svensson et al. Biochem Soc Trans 2007, 35, (Pt 3), pp 588-93. Read more |
|